DENTAL 3D PRINTING
Materials & Best Practices

A “3D printing fever” has taken over the dental industry and it’s not a surprise, considering that permanent restorations can be produced with a material input as low as $2. Behind these innovations are companies committed to high-quality biocompatible 3D printing results with every print, such as BEGO and Ackuretta.
While the equipment has become increasingly easy to use for any dental professional, the need for a thorough education on the “How to use…” front has become even more important. Thomas Kwiedor, Head of Business Development for 3D Printing at BEGO, joined us for a conversation discussing some of the most frequently asked questions about 3D printing materials and the proper post-printing procedures.
1. Mr. Kwiedor, I get a lot of questions about the calibration and validation of various materials. My favorite one so far was the following: “Why do we need to buy a curing oven? We can keep it in the sun and it will cure in 15 min, right?”
As much as I love the sun, and despite the tremendous power and all the positive effects that come from sunlight – "natural healing" is not indicated in the context of dental applications.
Companies, such as Ackuretta, sell medical-grade UV ovens because the curing has a significant impact on the physical properties of a dental application and therefore also on the biocompatibility.
To ensure reproducible results for medical products in terms of patient safety, all components for the manufacturing of dental restorations (including post-processing) are coordinated and tested – a time-intense process, which should be performed and overseen by experts. For this reason, only the recommended light-curing devices and the material-specific instructions of the manufacturers on the respective curing times should be used.
2. Another commonly asked question here: “I can calibrate a resin within 15 minutes in my office. Why would I pay to have a company do this process for me? Why do I need to validate a resin?"
We should perhaps illuminate the difference between calibration and validation.
In contrast to calibration, i.e. the measurement and, if necessary, adjustment of the printer, the verification, and validation of a 3D printing resin for dental applications involves much more than just calibration. Extensive tests such as tooth fit tests or biocompatibility tests, etc. under specified conditions (e.g., temperature) are part of the process, as is ensuring the reproducibility of the results. To meet the legal requirements and all standard requirements, the manufacturers spend weeks and months to fulfill the extensive requirements for obtaining product approval.
3. The 3D printing world is full of abbreviations that aren’t necessarily intuitive. For example, what does IFU stand for?
IFU is the abbreviation for Instructions for Use. IFU means the information provided by the manufacturer informing the user of the intended purpose and correct use of a product and any precautions to be taken.
For example, when we talk about IFUs for 3D printing resins, they also include basic information about the handling of equipment and materials required to produce an approved (medical) product, i.e. the dental restoration made from the resin. Instructions for use for medical devices are subject to strict regulatory requirements as part of the product identification (labeling).
If you use 3D printers in your office, you should ensure that you are aware of the IFUs of all your equipment.
4. Many customers don’t like the IPA. I’ve been asked whether your resins are water washable, what’s your answer to that?
Whether it is possible to clean the printed objects with water or with IPA (Isopropyl Alcohol) depends on the chemical composition of the resins. With regard to our 3D printing materials, IPA has proven to be a reliable cleaning agent and leaves a clean solvent-free surface after cleaning, which is beneficial to product quality. Of course, the instructions for use should be followed and IPA should not be handled with bare hands.
5. Mr. Kwiedor, I often get told by customers that they believe, they can do their own “validated workflow”. What would you respond to that statement?
Your customers are absolutely right with this statement. Is that why I would recommend that they do it?
The answer is no.
Before joining BEGO more than 11 years ago, I myself worked for many years as the managing director of large laboratories and am also a qualified auditor.
I am therefore very familiar with the regulatory requirements for medical devices, which have increased significantly in recent years. These requirements serve patient safety and are monitored by the respective authorities. It’s a multi-tiered approval process that can take years to fulfill.
So why should users invest significant time and financial resources to release validated processes when their industry partners already offer them validated solutions and the corresponding assurance to meet the necessary requirements.
6. DIY printers come without calibration and the power supply is often unstable. Do you see a danger in users calibrating BEGO resin and getting hit-and-miss results?
Yes, we do see a great risk in those processes, which is why we only recommend qualified 3D printers, such as the Ackuretta SOL and DENTIQ.
































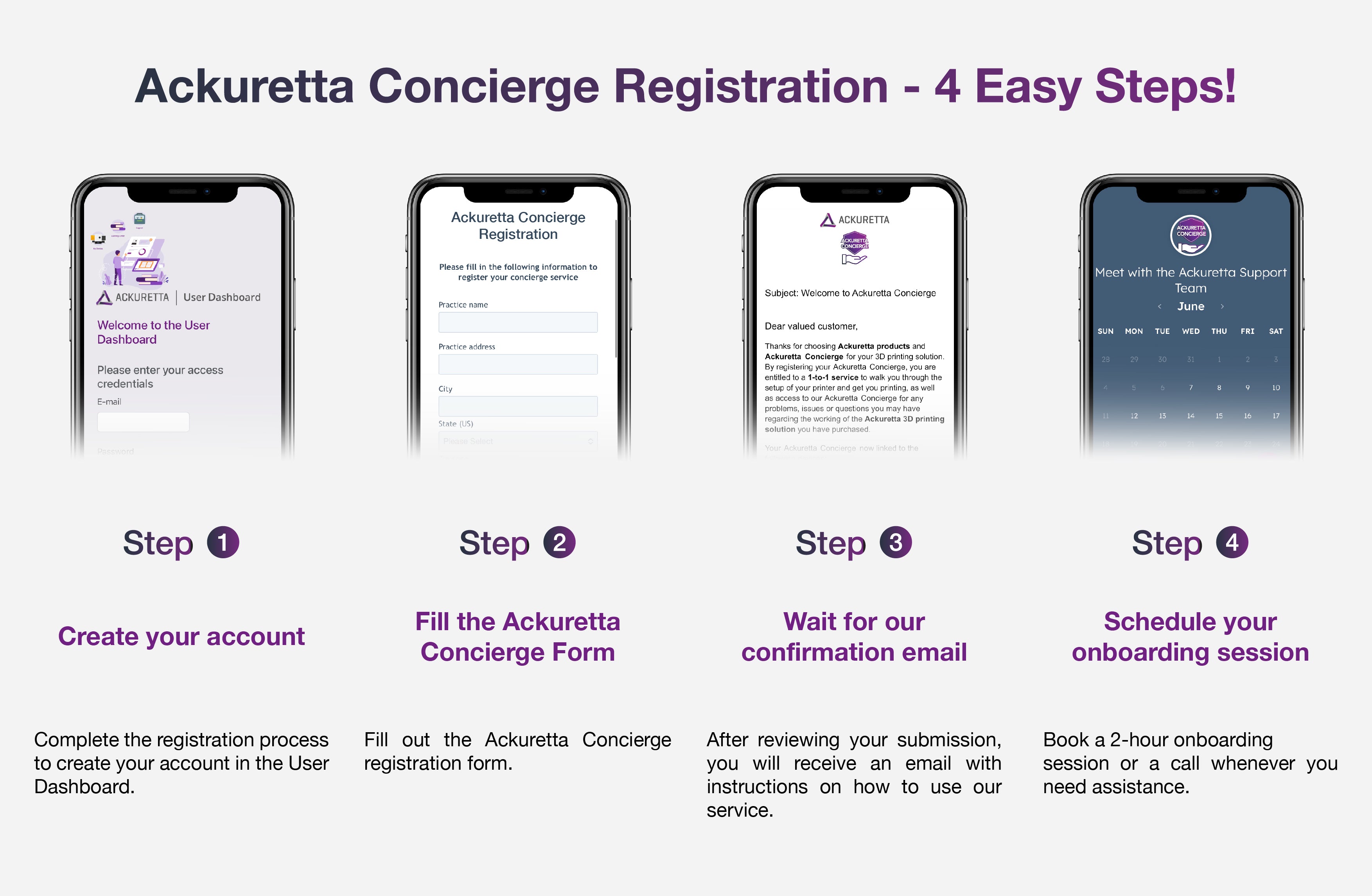






























 Loading ...
Loading ...








